TheraB Medical: SnugLit™Wearable Phototherapy System
TheraB Medical: SnugLit™
Wearable Phototherapy System
The Bonding Breakthrough: How Cortex Design steered TheraB Medical from a senior thesis to FDA 510(k) clearance and a successful acquisition in just 30 months.

TheraB Medical: SnugLit™ Wearable Phototherapy System
The Bonding Breakthrough: How Cortex Design steered TheraB Medical from a senior thesis to FDA 510(k) clearance and a successful acquisition in just 30 months.
Reframing Phototherapy Around the Infant
For decades, treating neonatal jaundice meant one thing: isolation. High-intensity blue-light systems required infants to remain tethered to stationary bassinets, physically separating families during the most critical window of early development.
TheraB Medical envisioned a radical alternative: SnugLit™, a wearable system that turned the treatment into a swaddle. The goal was to maintain clinical efficacy while allowing parents to hold, feed, and bond with their newborns.
What began as a senior engineering thesis quickly collided with the “Valley of Death” in medtech. For TheraB’s solo founder and CEO, Alexa Jones, the leap from academic concept to regulated medical device was fraught with existential risks. A single technical misstep or regulatory dead end could have shuttered the company before its first sale.
Cortex Design didn’t just take a brief; they took a stake. Joining as a strategic partner and direct investor, Cortex assumed the role of an integrated product development arm. By aligning their success with TheraB’s commercial viability, Cortex provided the “Fractional CTO” leadership a solo founder requires to move fast.
From system architecture and industrial design to the gruelling nuances of ISO 13485-certified quality systems, Cortex provided the decision-making authority to navigate the 510(k) pathway. The result? A move from concept to clearance in roughly two and a half years—a blistering pace for a novel Class II medical device.

TL;DR
- The challenge: Traditional neonatal phototherapy is clinically effective but it disrupts the bonding between infants and caregivers. It also does not adapt easily to environments outside hospitals. Developing a wearable alternative involved significant technical, regulatory, and commercial risks, especially for a first-time founder.
- Cortex’s role: Cortex Design provided end-to-end product leadership, acting not as an outsourced engineering firm, but owning system architecture, industrial design, design engineering, embedded systems, human factors, design controls and outsourced regulatory strategy, while directly investing in TheraB Medical.
- What made the difference: Fractional CTO Leadership, early product de-risking, design-led human-centred insights across hospital and home workflows, integrated design controls from day one, and capital-efficient prototyping/ manufacturing decisions that enabled a goggles-free wearable phototherapy system.
- The outcome: A production-ready, FDA 510(k)-cleared wearable phototherapy system delivered in approximately two and a half years, clinically robust, intuitive to use, and designed to preserve infant–caregiver bonding during the first days of life

“Cortex was instrumental in helping us take SnugLit from early prototype to a 510(k)–cleared device. Building a physical product medical device startup is demanding, and having the right development partner can make or break the journey. Cortex truly felt like an extension of TheraB, and their technical expertise helped bring both the product and the business to a meaningful inflection point.”
-Alexa Jones, Founder, TheraB Medical
On site early design exploration session with client

Designing for the Hospital and the Home
Traditional phototherapy for neonatal jaundice often relies on stationary equipment that limits infant mobility and caregiver interaction. SnugLit™ was conceived as a wearable phototherapy system, designed from the start to deliver effective light therapy in a portable form that could support infant care across both hospital and home settings. Achieving that continuity without introducing new safety or workflow risk was an early, non-negotiable requirement for the product’s clinical and commercial viability.
Failure to support continuity between hospital and home care would have created material risk on multiple fronts: low clinician adoption due to workflow friction, increased caregiver misuse outside controlled environments, and regulatory exposure driven by reliance on training rather than design controls. From a business perspective, any breakdown in this transition would have limited SnugLit™ to niche inpatient use, undermining the commercial rationale for a wearable system.

Most neonatal devices are optimized for a single, tightly controlled environment. SnugLit™ had to function across two fundamentally different contexts: the hospital nursery and the home.
Hospitals demand speed, reliability, and minimal cognitive overhead. Home use introduces variability; charging behaviour, cleaning practices, and caregiver experience levels, all while maintaining safety and compliance.
Cortex conducted boots‑on‑the‑ground ethnographic research with neonatal nurses, clinicians, and expectant parents to understand how phototherapy is actually delivered, interrupted, resumed, cleaned, and monitored across settings. These insights directly shaped system architecture decisions, including how treatment time is tracked, how components are swapped, and how misuse is prevented without reliance on training alone.
Designing for continuity between hospital discharge and home care elevated SnugLit™ from a single device into a portable phototherapy system that supports continuity of care beyond the hospital.
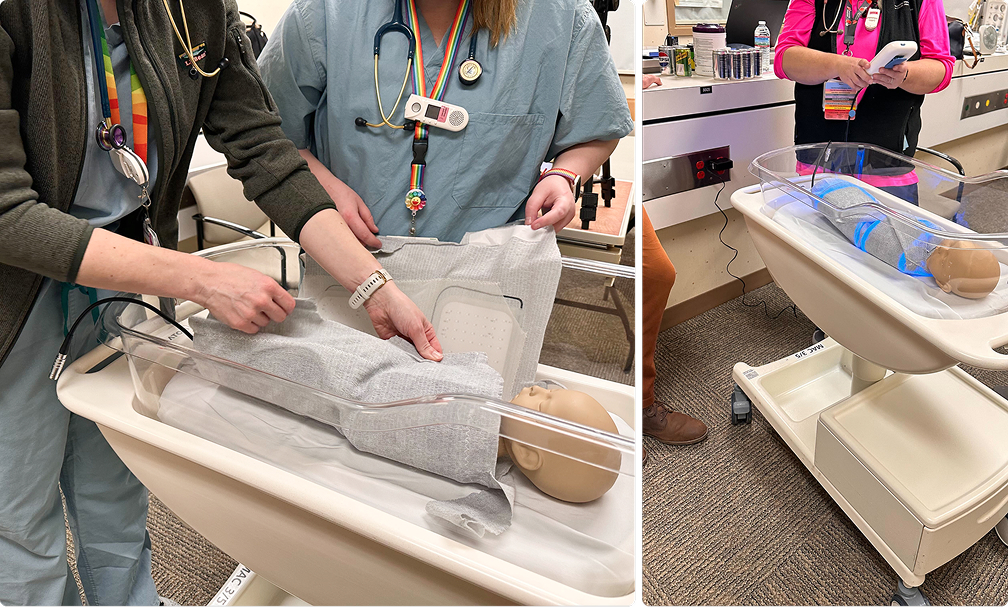
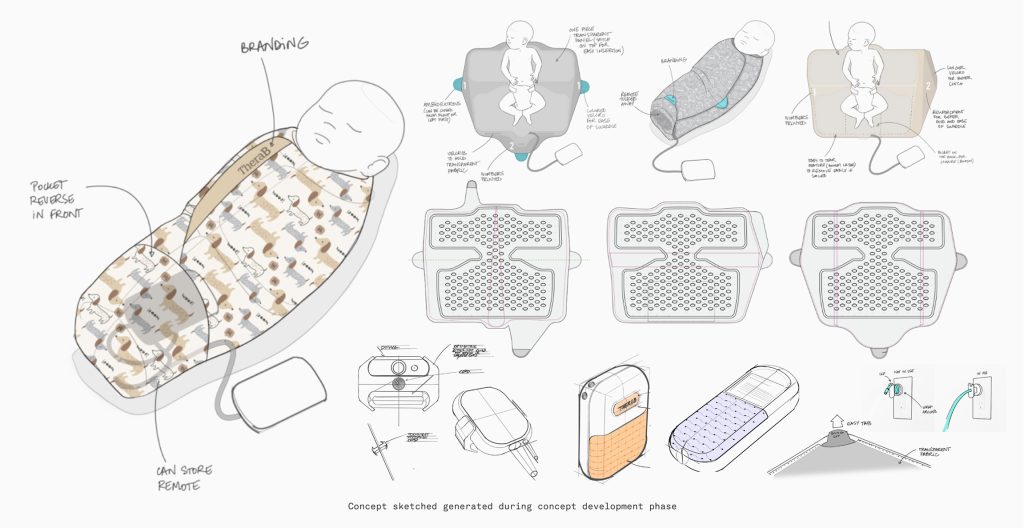
Early Derisking: Is This Worth Building?
Early in the program, Cortex made an explicit, irreversible architectural decision: the system would not rely on active cooling. This was not the outcome of late‑stage testing, but a deliberate product call made up front, based on the clinical, commercial, and regulatory consequences of introducing moving parts into a neonatal wearable device.
Before committing to full development, Cortex ran a focused technical derisking phase, BaseCamp by Cortex, to pressure‑test the assumptions most likely to end the program or materially extend the timeline and development budget if they proved false.
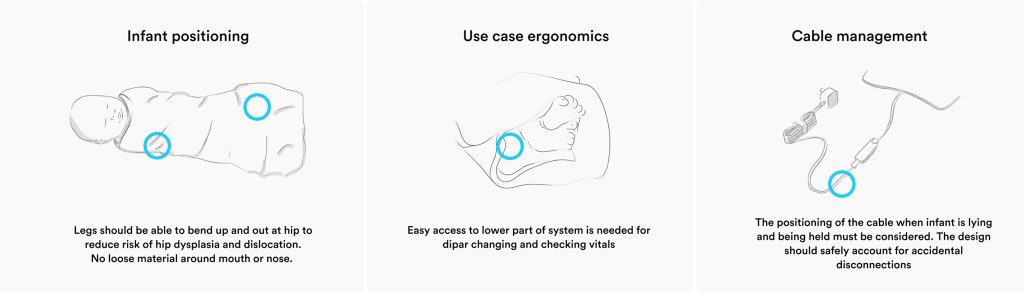
Preliminary design findings uncovered in Basecamp
Three assumptions rose quickly to the top. First, that the system could deliver phototherapy at the clinically optimal wavelength, centred around 475 nm, using commercially viable blue LEDs, without compromising output consistency or lifetime. Second, that thermal performance could be safely managed despite wrapping the infant in a swaddle, where conventional thermal modelling breaks down, and excess heat becomes a direct patient‑safety risk.
The third assumption was inseparable from the first two. An earlier concept proposed an active cooling fan. Cortex rejected that approach outright. Introducing a fan would have increased cost, noise, and contamination risk—and in a system designed to wrap an infant, it forced the team to confront the very real question of what happens when shit literally hits the fan.
An Immersion Circulator (Sous-Vide) was used to mimic infant body temperature to validate the system’s passive cooling.

Rapid in‑house testing challenged these assumptions directly, including airflow, thermal behaviour, and cleanability scenarios that standard design reviews often overlook. With active cooling eliminated, the team focused on reducing heat at the source—selecting high‑efficiency blue light LEDs and intentionally minimizing drive current to achieve the required irradiance with lower thermal load. This combination of component selection and electrical design validated that the system could operate safely without forced air cooling, avoiding the long‑term complexity and maintenance burden of fans and preventing scenarios as unglamorous as scraping poop off a fan blade.
These questions were resolved early, with minimal prototype investment, allowing later development phases to proceed with fewer downstream surprises.
Designing for Real-World Use
Clinical Efficiency Meets Caregiver Confidence
In practice, hospital nurses need devices that deploy quickly and recover gracefully from interruptions. SnugLit™ was designed so Controllers could be swapped without losing treatment time, shifting session tracking to the Light Mat itself and reducing downtime when batteries run low.
For parents and caregivers, clarity and reassurance matter more than throughput. Interface language mirrors clinical prescriptions, cleaning protocols are explicitly home‑safe, and the disposable Swaddle reduces anxiety around hygiene and setup.

Engineering Out a Known Safety Problem
Traditional phototherapy relies on eye masks, a long‑standing safety control with well‑documented failure modes driven by dislodgement, inconsistent placement, and reliance on vigilance rather than design. In high‑stress neonatal environments and during home use, these procedural controls are fragile, vulnerable to fatigue, movement, and caregiver variability.
Very early in concept development, Cortex recognized that external eye protection, assumed by legacy phototherapy standards, would be fundamentally incompatible with a wearable system intended for prolonged use, caregiver handling, and home use. Rather than accepting goggles as a fixed requirement, eye safety was treated as a system‑level industrial design problem to be solved during conceptualization.
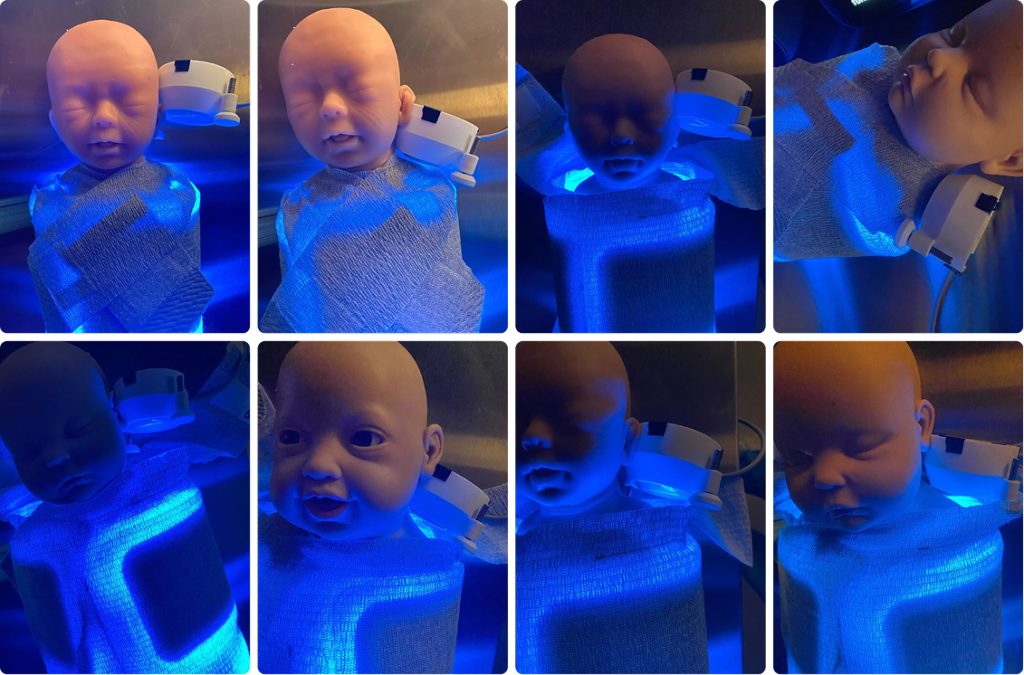
SnugLit™ replaces that paradigm with light blocking integrated directly into the Swaddle. The textile shielding blocks errant light at the source, eliminating the need for separate eye masks and removing a critical failure mode through intuitive design rather than instruction.
To confirm the validity of the design, Cortex executed formative usability studies, recruiting and testing with both nurses and lay caregivers across hospital and home contexts. This included iterative formative and pilot studies led by Cortex to identify and mitigate use-related risks prior to summative validation. These studies informed labelling, iconography, swaddling cues, and system behaviours, ensuring correct use in high‑stress care scenarios. The system was intentionally tolerant of varying experience levels, reducing the likelihood of use errors and minimizing patient risk.

“At Cortex, we don’t just design for compliance; we design for connection. With TheraB, the challenge wasn’t just hitting 475 nm of blue light—it was ensuring a mother could hold her child during that treatment. Taking the lead on the totality of this 510(k) development allowed us to prove that technical rigor and human empathy are not mutually exclusive. We didn’t just build a device; we helped Alexa build a company.”
-Andrew Lowe, Partner & Sr. Director of Product, Cortex Design
From Concept to Manufacturable System
Iteration Without Excess Capital
Rather than chasing polished early prototypes, Cortex prioritized rapid learning and system design confirmation. In‑house mechanical testing, 3D‑printed assemblies, and parallel flex‑PCB variants allowed side‑by‑side evaluation of architectures before design freeze. This approach compressed timelines while preserving flexibility to adapt to any development challenges.

Flexible Electronics at Production Scale
Delivering uniform, wrap‑around phototherapy required embedding a custom flexible PCB within a medical‑grade silicone matrix. That decision carried consequences. The challenge was durability: repeated bending, thermal cycling, and cleaning without delamination or solder fatigue.
The construction approach was a deliberate strategic decision by Cortex: defer tooling investment to preserve iteration speed and reduce capital risk.
Cortex developed and validated a silicone‑encapsulation process that protected electronics while maintaining flexibility. Importantly, the flex PCB was laminated between two layers of liquid silicone rubber using adhesive, a construction method intentionally chosen because it could be executed at low volume without investing in compression moulds or LSR overmolding tooling. This allowed the team to refine geometry, materials, and routing between prototype revisions without incurring large capital costs. Accelerated lifecycle testing, using custom fixtures to simulate thousands of bending cycles, confirmed long‑term reliability before committing to tooling.
In-house crease testing
Design Decisions driven by Risk Management
Safety was embedded at the hardware and firmware level: thermal monitoring, automatic shutdowns, and mechanical light containment—supported by the system’s low-heat optical and electrical design—reduced reliance on user behaviour. This approach simplified human factors validation by demonstrating that critical risks were controlled by design, not documentation. Pre‑compliance testing and internal validation surfaced issues early, reducing uncertainty during formal evaluations and regulatory review.
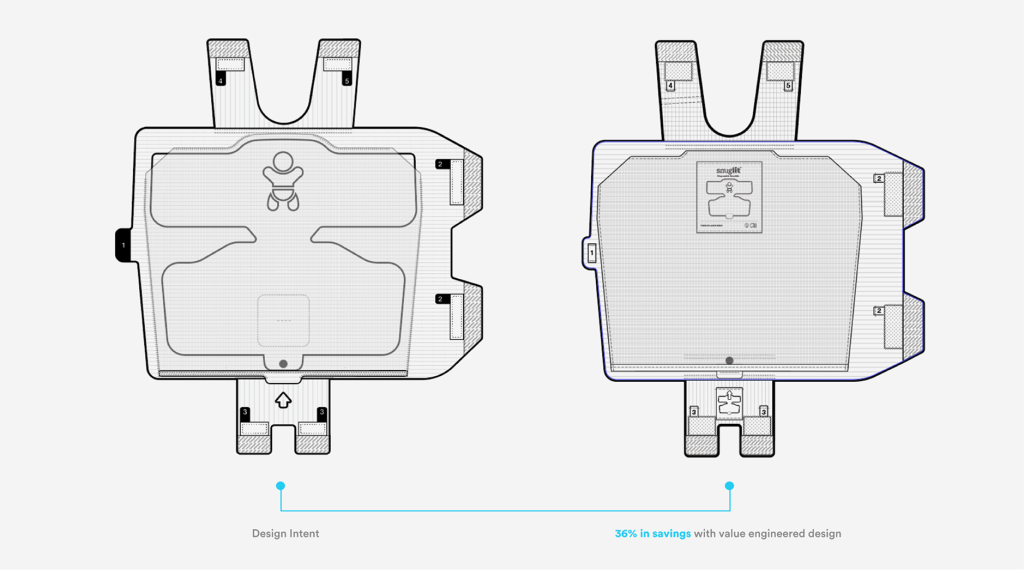
Cost Discipline Without Compromise
Throughout development, design decisions were evaluated against cost‑of‑goods targets. Close collaboration with suppliers enabled material and process optimizations that reduced cost while preserving safety margins, performance, and regulatory intent.
Designing for Regulation, Not Around It
Design Controls Built In from Day One
Regulatory success demanded more than standards interpretation. Cortex assumed responsibility for the design controls infrastructure required to withstand scrutiny. From the earliest phases of the program, Cortex managed the Design History File (DHF) and Risk Management File within its ISO 13485:2016–certified Quality Management System, keeping the program audit‑ready throughout development.
Design controls were integrated, not deferred. Conceptual design informed early requirements, which then drove engineering, verification, and validation activities, including human factors engineering. Cortex managed third‑party compliance testing across core IEC 60601 standards (including electrical safety, EMC, and alarms) and authored the summative usability protocol, supporting independent execution while overseeing study readiness, execution, and remediation. This approach preserved creative flexibility early while introducing regulatory formality as required, eliminating a common failure mode for first‑time founders: discovering late that design documentation cannot withstand FDA review.
Pilot Usability testing conducted in-house at Cortex Design

The international standard governing phototherapy devices, IEC 60601‑2‑50, was written for stationary systems built around a single overhead light source, not a wrap‑around light mat containing hundreds of individual blue light LEDs. Applying that standard to SnugLit™ introduced a non‑trivial technical challenge: how to interpret requirements for irradiance measurement and averaging in a system with 455 discrete LEDs distributed across the infant’s body.

Cortex addressed this challenge in close collaboration with a regulatory consultant who brought prior, hands‑on experience with infant phototherapy devices and FDA 510(k) submissions. Rather than treating the standard as a box‑checking exercise, the combined team focused on its underlying clinical intent and developed defensible, intuitive methods for characterizing light output that regulators could clearly understand and trust.

Outcome
The collaboration ultimately produced a production‑ready, FDA 510(k)‑cleared wearable phototherapy system shaped by a deliberately human‑centred design approach. By solving for both hospital and home workflows from the outset, Cortex helped TheraB Medical deliver a device that clinicians could deploy quickly, caregivers could use intuitively, and families could trust during an emotionally intense period of care.
SnugLit™ eliminates the need for infant eye goggles through integrated light containment, reduces setup and monitoring burden through workflow‑aware system architecture, and enables parents to hold, feed, and bond with their newborns during treatment. Together, these decisions produced a clinically robust phototherapy system that preserves infant–caregiver bonding during the first critical days of life and shows how rigorous human‑centred design, applied in a regulated environment, can materially improve both the care experience and the commercial viability of a medical product.


Services provided
- QMS management
- Design Controls
- Industrial Design
- Packaging Design
- UX/UI design
- Embedded Design
- Regulatory Implementations
- Compliance testing
- Formative usability testing
- Risk Management
- Design Verification
- Mechanical design
- Design for manufacturing
- Benchtop testing
- Labeling development
- Supply chain sourcing
- Compliance testing
- Manufacturing transfer
- QMS management
- Design Controls
- Industrial Design
- Packaging Design
- UX/UI design
- Embedded Design
- Regulatory Implementations
- Compliance testing
- Formative usability testing
- Risk Management
- Design Verification
- Mechanical design
- Design for manufacturing
- Benchtop testing
- Labeling development
- Supply chain sourcing
- Compliance testing
- Manufacturing transfer
From design to FDA clearance to acquisition, our journey has been defined by partnership. Cortex wasn’t simply a collaborator, they were a true partner, standing shoulder to shoulder with TheraB at every milestone. They championed the mission, invested in the vision, endured the challenges, and demonstrated an unwavering commitment to ensuring SnugLit reached the patients who needed it most.
-Chris Mathia, CEO, TheraB Medical
